 |
| The Chimera of Arezzo, an Etruscan effigy of the monster |
How is a mantidfly akin to the Chimera–a monstrous fire-breathing beast of Greek myth
 |
| A Louisianan C. brunnea, photographed by Gayle Strickland |
 |
| Lomamyia sp., the only genus of beaded lacewing found in the United States; picture by Randy Hardy |
 |
| A Plega sp. (Symphrasinae) making rude tarsal gestures at the photographer (Marcela Freerks) |
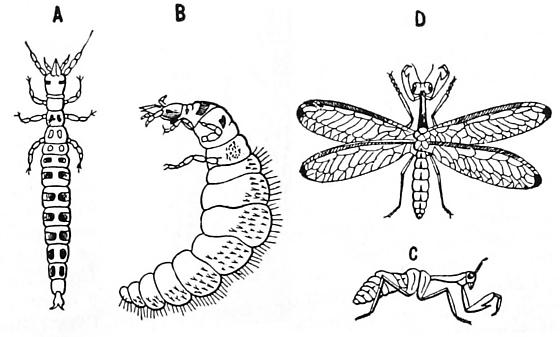 |
| The hypermetamorphic stages of Mantispa sp., as shown in Introduction to Entomology (1933); not to scale |
 |
| A first-instar mantispine situated near a jumping spider's (Salticidae) pedicel; photograph by Jeff Kramer |
 |
| Ocnaea sp. (Acroceridae), female, photographed by Jeff Gruber |
Happy New Year (if it is a happy one–which I doubt).
*That is, hitchhiking, as opposed to parasitic.
_________________________________________________________________
Buys, S. C. (2008). Observations on the biology of Anchieta fumosella (Westwood 1867) (Neuroptera: Mantispidae) from Brazil. Tropical Zoology, 21, 91-95.
Drees, B. M. and Jackman, J. (1999). Field Guide to Texas Insects. Houston: Gulf Publishing Company.
Enderlein, G. (1910). Klassifikation der Mantispiden nach dem Material des Stettiner Zoologische Museums. Stettiner Entomologische Zeitung, 71, 341-379.
Gordh, G. and Headrick, D. H. (2003). A Dictionary of Entomology. Wallingford: CABI.
Hook, A. W.; Oswald, J. D. and Neff, J. L. (2010). Plega hagenella (Neuroptera: Mantispidae) parasitism of Hylaeus (Hylaeopsis sp.) (Hymenoptera: Colletidae) reusing nests of Trypoxylon manni (Hymenoptera: Crabronidae) in Trinidad. J. Hymenopt. Res., 19, 77-83.
Imms, A. D. (1957). A General Textbook of Entomology. London: Methuen.
Jarzembowski, E . A. (1980). Fossil insects from the Bembridge Marls, Palaeogene of the Isle of Wight, southern England. Bulletin of the British Museum of Natural History (Geology), 33, 133-140.
Johnson, J. B. and Hagen, K. S. (1981). A neuropterous larva uses an allomone to attack termites. Nature, 289, 506-507.
Lambkin, K. J. (1986). A revision of the Australian Mantispidae (Insecta: Neuroptera) with a contribution to the classification of the family I. General and Drepanicinae. Australian Journal of Zoology (Supplementary Series), 116, 1-142.
Maia-Silva, C.; Hrncir, M.; Koedam, D.; Machado, R. J. P. and Imperatriz-Fonseca, V. L. (2012). Out with the garbage: the parasitic strategy of the mantisfly Plega hagenella mass-infesting colonies of the eusocial bee Melipona subnitida in northeastern Brazil [electronic version]. Naturwissenschaften, 100, 101-105. Retrieved 1/6/13 from http://link.springer.com/content/pdf/10.1007%2Fs00114-012-0994-1
Makarkin, V. N. (1990). Novye setchatokrylye (Neuroptera) iz verkhnego mela Azii. In: I. A. Akimov (ed.), Novosti Faunistiki I Sistematiki (pp. 63-68). Kiev: Naukova Dumka.
Parfin, S. I. (1958). Notes on the bionomics of the Mantispidae (Neuroptera: Planipennia). Entomological News, 69, 203-207.
Redborg, K. E. (1998). Biology of the Mantispidae. Annual Review of Entomology, 43, 175-194.
Redborg, K. E. and MacLeod, E. G. (1983). Climaciella brunnea (Neuroptera: Mantispidae): a mantispid that obligately boards spiders. Journal of Natural History, 17, 63-73.
Tjeder, B. (1959). Neuroptera-Planipennia. The lace-wings of southern Africa. 2. Family Berothidae. In: Hanström, B.; Brinck, P. and Rudebec, G. (eds.), South African Animal Life Results of the Lund University Expedition in 1950-51 (vol. 6, pp. 256-314). Stockholm: Almqvist and Wiksell.
Wedmann, S. and Makarkin, V. N. (2007). A new genus of Mantispidae (Insecta: Neuroptera) from the Eocene of Germany, with a review of the fossil record and biogeography of the family [electronic version]. Zoological Journal of the Linnaean Society, 149, 701-716. Retrieved 1/6/13 from http://www.biosoil.ru/files/00004954.pdf
Willmann, R. (1990). The phylogenetic position of the Rhachiberothinae and the basal sister-group relationships within the Mantispidae (Neuroptera). Systematic Entomology, 15, 253-265.
No comments:
Post a Comment